Monepantel
The Current Treatment Landscape
The current standard of care treatment is multiagent chemotherapy with CHOP (cyclophosphamide, hydroxydaunorubicin, vincristine and prednisone) the most frequently adopted protocol. While CHOP has a high response rate, it has significant side effects ranging from gastrointestinal disturbance to bone marrow suppression. Chemotherapy is a high-cost treatment with poor quality of life outcomes, and poses major safety risk to owners handling pets.
Monepantel Studies
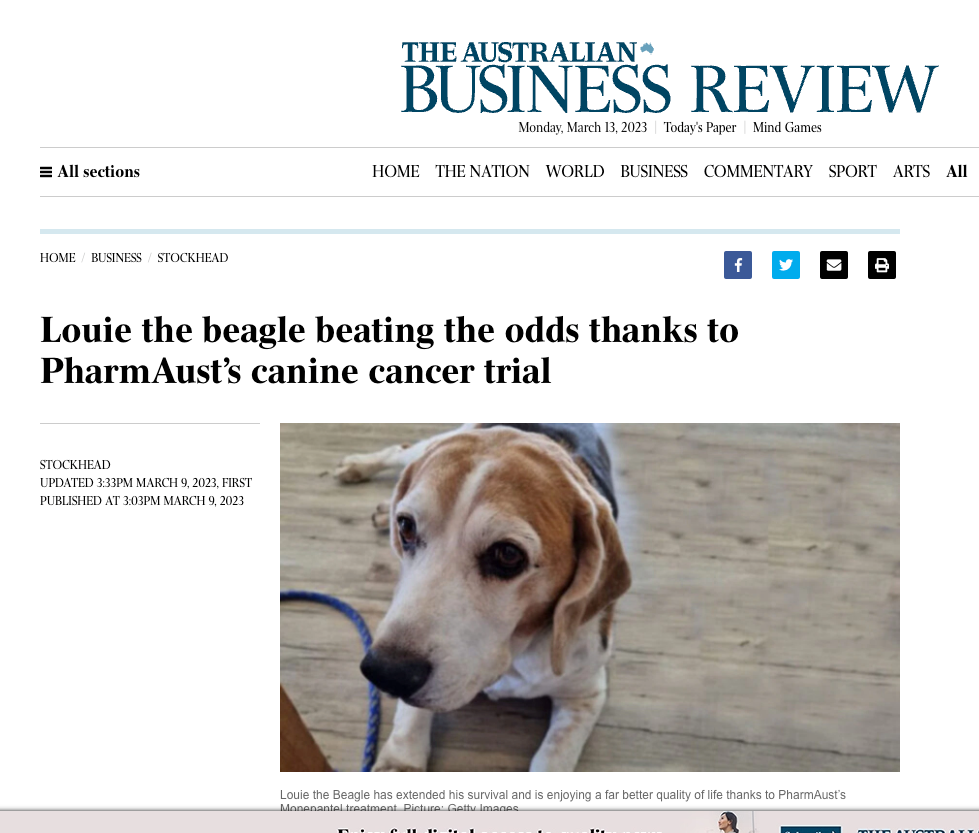
The Phase 2 study added to monepantel’s safety profile established in a Phase 1 study with minimal side effects and no reported adverse events. Significantly, monepantel consistently demonstrates a significant competitive advantage in quality of life (QoL) and level of function (LoF) when compared to existing treatments.
Following promising Phase 2 results, PharmAust is preparing to initiate a field trial for product registration.
Attractive Commercial Opportunity
An estimated 6 million dogs are diagnosed with cancer each year in the US compared to 1.7 million humans. Many of the most common cancers in dogs (eg. lymphoma, osteosarcoma, mammary) are recognised as being very similar to human cancers. As such, dogs are increasingly used as models for human disease.
PharmAust has shown that monepantel is a cost-effective, safe and potent therapy which meets the unique combination of criteria required to generate a commercial return in the companion animal drug market.